Rituximab and B-Cell Therapy Can Delay Lyme Disease Diagnosis
B-cell suppressive therapies such as rituximab can delay Lyme disease diagnosis. These medications reduce the body’s ability to produce antibodies — the same antibodies that standard Lyme disease tests rely on to detect infection. As a result, patients receiving immune-modulating therapy may have false-negative or borderline Lyme disease tests despite active infection.
This case illustrates how immune suppression can interfere with Lyme disease testing and diagnosis.
I will be discussing a 20-year-old woman with a three-year history of multiple sclerosis who was treated with B-cell suppressive therapy. She was prescribed two biological disease-modifying antirheumatic drugs (DMARDs).
The case was reported by Sjöwall and colleagues in the journal Frontiers in Neurology.
Immune-Modulating Therapy for Multiple Sclerosis
The patient was initially treated with tocilizumab, marketed in the United States as Actemra. Tocilizumab is a monoclonal antibody that blocks signals from the interleukin-6 (IL-6) receptor.
This medication is commonly used for autoimmune conditions such as rheumatoid arthritis. While it can reduce inflammation, it can also lower the immune system’s ability to fight infections.
The patient was later prescribed rituximab, sold under the brand name Rituxan. Rituximab is another monoclonal antibody that targets CD20 on B-cells and suppresses B-cell activity.
Because B-cells produce antibodies, suppressing them can interfere with the body’s normal immune response to infection.
Lyme Disease Symptoms During B-Cell Therapy
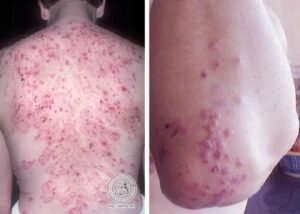
The patient developed knee arthritis and a persistent rash that lasted for six months.
Although Lyme disease was considered, the diagnosis was dismissed because Lyme disease tests were negative.
However, the negative test results were misleading. B-cell suppressive therapy can prevent patients from producing the antibodies that standard Lyme disease tests detect.
Delayed Treatment for Lyme Disease
The patient was eventually treated with doxycycline six months after her symptoms began.
This case highlights an important clinical lesson: Lyme disease testing may be unreliable in patients receiving B-cell suppressive therapy.
When immune-modulating medications suppress antibody production, clinicians may need to rely more heavily on symptoms, exposure history, and clinical judgment rather than antibody testing alone.
Clinical Insight
Standard Lyme disease testing depends on antibody production. When medications such as rituximab suppress B-cells, patients may not produce detectable antibodies even when infection is present. In these situations, Lyme disease can be overlooked or diagnosed late.
Reference
Sjöwall J, Xirotagaros G, Anderson CD, Sjöwall C, Dahle C. Case Report: Borrelia-DNA Revealed the Cause of Arthritis and Dermatitis During Treatment With Rituximab. Front Neurol. 2021;12:645298.
You can hear more about these cases through Dr. Cameron’s blogs, social media, and YouTube channel. Sign up for the newsletter to stay updated on new cases and research.
How to Connect with Dr. Daniel Cameron:
- Website: https://www.DanielCameronMD.com
- Call his office: 914-666-4665
- Email: DC******@*************MD.com
- Contact form: Contact Dr. Cameron
- Instagram: Instagram
- Facebook: Facebook
- Twitter/X: Twitter
- YouTube: YouTube Channel
Please remember that the advice given is general and not intended as specific advice for any individual patient. If you require specific medical advice, consult an experienced healthcare professional.
Dr. Daniel Cameron, MD, MPH
Lyme disease clinician with over 30 years of experience and past president of ILADS.
Symptoms • Testing • Coinfections • Recovery • Pediatric • Prevention