Central Sensitization in Lyme Disease
Some Lyme disease patients become unusually sensitive to pain, sound, light, or touch.
Brain imaging studies suggest abnormal sensory processing may occur.
These changes may contribute to fatigue, pain, and sensory overload.
Studies have found that patients with Lyme disease (LD) experience “exaggerated responses to pain and non-painful stimuli” despite antibiotic treatment. [1,2] Lopez-Sola and colleagues describe similar responses in patients with fibromyalgia (FM), a condition associated with widespread pain, fatigue, cognitive symptoms, sleep problems, and sensory hypersensitivity. [3]
In addition to pain-related changes, fibromyalgia patients may also demonstrate reduced tolerance to non-painful sensory stimulation involving light, sound, smell, touch, and temperature. [3] These findings were reported in the journal PAIN, published by the International Association for the Study of Pain.
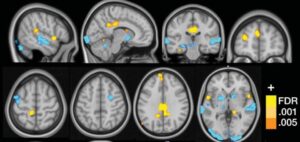
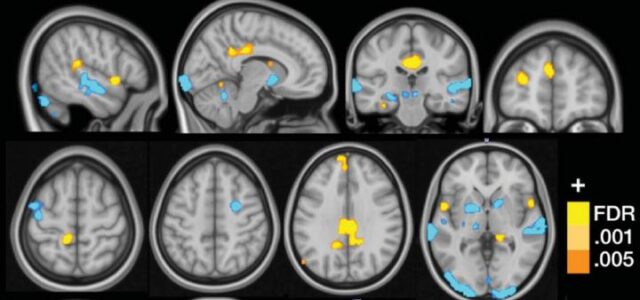
Brain Imaging and Exaggerated Sensory Responses
Lopez-Sola’s team used functional Magnetic Resonance Imaging (fMRI) to demonstrate exaggerated responses to both painful and non-painful sensory stimuli in fibromyalgia patients. [3]
Functional MRI has previously been used to measure regional changes in brain metabolism and neural activity associated with disease, treatment response, and pharmacologic effects. [4] PET and SPECT scans are also used to study brain function.
The researchers exposed 37 fibromyalgia patients and 35 control patients to painful pressure along with non-painful visual, auditory, and tactile stimulation.
Fibromyalgia patients demonstrated greater Neurologic Pain Signature (NPS) responses during painful stimulation, along with amplified responses to non-painful sensory input.
“In fibromyalgia, the misfiring and irregular engagement of different parts of the brain to process normal sensory stimuli like light, sound, pressure, temperature and odor results in pain, flu-like sensations or other symptoms,” explains Jan Chambers of the National Fibromyalgia & Chronic Pain Association. [5]
Central Sensitization and Lyme Disease
Patients with Lyme disease frequently describe symptoms consistent with central sensitization or sensory hyperarousal.
Acute and chronic pain associated with Lyme borreliosis has been characterized as having “qualities of neuropathic pain,” including deep aching, lancinating pain, radicular pain, and sensory abnormalities. [2]
Persistent pain despite antibiotic treatment has also been reported in some neuroborreliosis patients. [2]
Batheja and colleagues reported that patients with post-treatment Lyme syndrome (PTLS) commonly experience sensory hyperarousal affecting hearing and vision. [1]
Some patients become hypersensitive to:
- sound
- light
- touch
- smells
- temperature changes
According to Batheja, some individuals become unable to tolerate normal conversation volume and may wear ear protection in everyday environments. [1]
Others report wearing sunglasses indoors or avoiding daylight because of heightened visual sensitivity. [1]
Amplified Sensory Processing
The fibromyalgia imaging studies suggest that abnormal sensory processing may occur in several brain regions involved in pain integration, multisensory processing, and self-referential activity.
Lopez-Sola described augmented responses involving the insula/operculum, posterior cingulate cortex, and medial prefrontal regions. [3]
The authors cautioned that exaggerated responses to pain and non-painful stimuli are not unique to fibromyalgia and may occur in multiple chronic pain conditions involving sensitization. [3]
Exaggerated responses to pain and non-painful stimuli described in Lyme disease may eventually be measurable through advanced imaging techniques such as fMRI, helping clinicians assess therapeutic approaches and predict treatment responses in individual patients.
Why These Findings Matter
Central sensitization may help explain why some Lyme disease patients experience persistent pain, sensory overload, fatigue, dizziness, or cognitive dysfunction despite normal routine testing.
These symptoms are often misunderstood because they may not produce obvious abnormalities on standard neurologic examinations or laboratory studies.
Recognizing sensory hyperarousal and abnormal sensory processing may help clinicians better understand the neurologic burden experienced by some patients with persistent Lyme disease symptoms.
References
- Batheja S, et al. Post-treatment Lyme syndrome and central sensitization. J Neuropsychiatry Clin Neurosci. 2013;25(3):176-86.
- Zimering JH, et al. Acute and chronic pain associated with Lyme borreliosis: clinical characteristics and pathophysiologic mechanisms. Pain. 2014;155(8):1435-8.
- Lopez-Sola M, et al. Towards a neurophysiological signature for fibromyalgia. Pain. 2016.
- Glover GH. Overview of functional magnetic resonance imaging. Neurosurg Clin N Am. 2011;22(2):133-9.
- Researchers Discover ‘Brain Signature’ for Fibromyalgia.
- Breakthrough First Step in Brain-Based Fibromyalgia Diagnosis.
- What’s That Smell?
Dr. Daniel Cameron, MD, MPH
Lyme disease clinician with over 30 years of experience and past president of ILADS.
Symptoms • Testing • Coinfections • Recovery • Pediatric • Prevention


