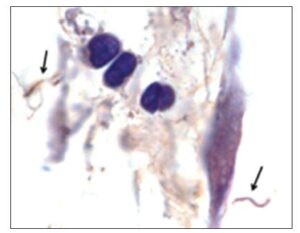
Persistent Lyme Infection and ‘Persister’ Cells
A key pattern is persistence. Some Lyme disease patients remain ill after treatment, raising questions about ongoing infection.
Experimental studies have demonstrated persistence of Borrelia burgdorferi across multiple animal models—including mice, rats, dogs, and non-human primates. :contentReference[oaicite:0]{index=0}
Some researchers suggest this may explain why a subset of patients continue to experience symptoms after treatment. In one analysis, as many as one-third of patients treated for Lyme disease remained chronically ill.
Start here: persistent symptoms after Lyme disease
What Are ‘Persister’ Cells?
A key pattern is antibiotic tolerance. Some bacteria can survive treatment without being resistant in the traditional sense.
In a review published in the Bosnian Journal of Basic Medical Sciences, Dr. Emir Hodzic describes ‘persisters’ as specialized bacterial cells that tolerate antibiotics.
“The main culprit responsible for the tolerance of pathogens to antibiotics is a specialized survivor – a persister,” states Hodzic.
Because persisters are dormant or slow-growing:
- They are not actively dividing
- They are difficult to culture
- They are less susceptible to antibiotics targeting active cells
This may help explain why symptoms can persist despite treatment.
How Borrelia Becomes Difficult to Eliminate
A key pattern is biological adaptation. Changes in the bacteria may reduce treatment effectiveness.
During infection, Borrelia burgdorferi can undergo plasmid loss—losing small DNA elements that affect growth and behavior.
These altered spirochetes:
- Remain viable
- Divide more slowly
- Become less responsive to antibiotics
This slower activity makes them more tolerant to antimicrobial therapy and harder to detect.
“There is clear scientific evidence that a small, heterogeneous subpopulation of surviving spirochetes shows tolerance to antimicrobial agents and can persist in a host for a prolonged period following therapy,” writes Hodzic.
Persistence Over Time
A key pattern is delayed reappearance. Persistent organisms may not be detectable immediately after treatment.
In animal models, non-cultivable spirochetes have been observed to reappear months after antibiotic therapy, including findings at 12 months post-treatment.
This delayed persistence may contribute to ongoing symptoms in some patients.
Why Standard Treatment May Be Less Effective Later
A key pattern is timing. Treatment appears more effective in early infection.
As infection progresses:
- Bacterial populations may change
- Persister cells may increase
- Treatment response may decline
This may explain why early treatment often leads to better outcomes than treatment initiated later.
Clinical Interpretation
A key pattern is incomplete resolution. Persistent symptoms may reflect more than a simple post-infectious process.
Hodzic notes that antibiotic tolerance differs from classic resistance. In these cases, multiple classes of antibiotics may fail to fully eliminate non-dividing or slow-growing organisms.
This raises important questions about ongoing symptoms and treatment response in Lyme disease.
Clinical Takeaway
Persistent forms of Borrelia burgdorferi may contribute to ongoing symptoms after treatment.
These ‘persister’ cells can survive antibiotics by entering a dormant or slow-growing state.
Understanding this pattern may help explain why some patients do not fully recover after standard therapy.
Dr. Daniel Cameron, MD, MPH
Lyme disease clinician with over 30 years of experience and past president of ILADS.
Symptoms • Testing • Coinfections • Recovery • Pediatric • Prevention